Breaking Insulating Barriers! Nat. Catal.: Dual-Atom Catalysts Reshape the "Electronic Properties" of High-Energy Solid-State Batteries
- Technical Research

- Apr 21
- 3 min read
Introduction: The "Insulation Passivation" Curse of Solid-Phase Conversions
In high-energy-density systems like Lithium-Sulfur (Li-S) and Lithium-Oxygen (Li-O₂) batteries, solid-phase conversion reactions are the primary energy source. However, driving these reactions to completion is notoriously difficult.
The core issue lies in "insulators." During discharge, the solid intermediate products generated (such as Li₂S₂ and Li₂O₂) are inherently insulating. They continuously deposit and coat the catalyst surface, acting as a wall that blocks electron conduction. Ultimately, the electrode surface is "passivated," reactions terminate prematurely, and the practical capacity falls far short of theoretical limits.
Recently, a collaborative team led by Prof. Feng Li, Prof. Zhenhua Sun (IMR, CAS), and Academician Hui-Ming Cheng (SIAT) published groundbreaking research in Nature Catalysis. They introduced a novel design paradigm that alters the "electronic properties" of intermediate products using Dual-Atom Catalysts (DACs), successfully turning insulators into "conductors."
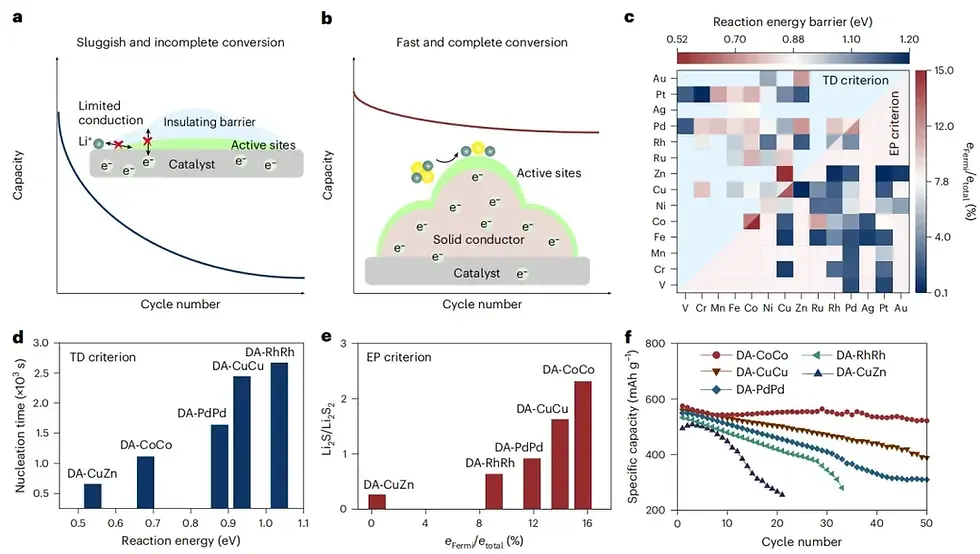
Paradigm Shift: From "Thermodynamics (TD)" to "Electronic Properties (EP)"
Historically, when designing catalysts, scientists have largely adhered to the "Thermodynamics (TD) first" principle—searching for materials with the lowest reaction energy barriers.
However, after conducting massive Density Functional Theory (DFT) calculations on 351 DACs, the research team discovered a counterintuitive phenomenon: Thermodynamics (low barriers) only dictate the initial nucleation stage. As intermediates accumulate, heteronuclear catalysts with TD advantages (like DA-CuZn) rapidly fail because the products are insulating.
The team proposed a brand new criterion: Electronic Properties (EP), focusing on the density of states near the Fermi level of the intermediate products. As long as the catalyst can endow the intermediates with conductivity, it will dominate the overall cycle life and conversion efficiency of the battery.

Unveiling Orbital Coupling: Homonuclear Dual-Atoms Make Insulators "Conductive"
Why do homonuclear dual-atom catalysts (like DA-CoCo) perform so exceptionally? The secret lies in "orbital symmetry" at the microscopic level.
Perfect Match & Dual-Way Electron Exchange: Homonuclear DA-CoCo possesses a symmetric πM* orbital distribution, which perfectly matches the symmetric σ* orbitals of insulating intermediates (like Li₂S₂). This alignment triggers a dual-way electron exchange.
Crossing the Fermi Level: Through this highly efficient orbital coupling, deep-level electrons in Li₂S₂ are activated, with approximately 15% of electrons shifting near the Fermi level (compared to just 0.2% for DA-CuZn). Macroscopically, the originally insulating Li₂S₂ layer miraculously attains ultra-high electronic conductivity.

Massive Active Site Migration: "Top-Level Deposition" Breaks the Passivation Curse
Endowing the insulating products with conductivity completely alters the internal electrochemical behavior:
Bottom-Level Deposition (DA-CuZn): The insulating layer accumulates, blocking electron pathways. Unconverted Li₂S₂ can only pile up at the restricted catalyst interface, causing the reaction to "die" rapidly.
Top-Level Deposition (DA-CoCo): Because the deposited lithium sulfide layer itself is conductive, the catalytic active sites successfully migrate from the "solid-solid interface" (catalyst-product) to the "solid-liquid interface" (product-electrolyte). Fresh conversion reactions can continuously occur directly on the surface of the existing products.

Macroscopic Validation: 459 Wh/kg Ampere-Hour Pouch Cells
The optimal catalyst, DA-CoCo, screened based on this EP criterion, demonstrated astonishing practical capabilities:
Under extremely harsh conditions (lean electrolyte, high areal loading), an ampere-hour scale (1.2 Ah) Li-S pouch cell achieved a staggering energy density of 459 Wh kg⁻¹ and cycled stably for over 30 rounds. Meanwhile, this catalyst also realized a highly efficient 4-electron conversion in Li-O₂ batteries, proving the universal applicability of the theory.

Conclusion & Outlook
This work not only refreshes the performance metrics of high-energy batteries but, more importantly, upgrades the foundational logic of catalyst design. It demonstrates that in energy storage systems involving massive solid-phase product generation, merely pursuing "low reaction barriers" is insufficient. "Endowing intermediates with conductivity" is the ultimate law to sustain catalytic vitality and break conversion barriers.
Literature Information
Tong Yu, et al., Breaking insulating barriers in solid-phase conversion reactions with dual-atom catalysts for high-energy lithium batteries, Nature Catalysis (2026). https://doi.org/10.1038/s41929-026-01525-8




Comments