Ending the Chaos: "Sequenced Interfacial Chemistry" Tames Lithium Metal Anodes via MgO Templating
- Technical Research

- Mar 24
- 3 min read
Introduction: The "Traffic Jam" at the Lithium Interface
Lithium metal (Li⁰) is considered the ultimate anode material due to its ultra-high theoretical capacity and lowest reduction potential. However, when lithium ions deposit on the electrode surface, they undergo a highly complex cascade of reactions: desolvation, nucleation, growth, and the formation of the Solid Electrolyte Interphase (SEI).
In traditional electrolyte systems, these reactions are highly coupled and severely chaotic in both time and space. It is much like a busy intersection without traffic lights—various components (ions, solvents, anions) collide randomly, making the interfacial behavior unpredictable. Macroscopically, this leads to uncontrollable dendrite growth, dead lithium generation, and a rapid drop in battery lifespan.
Recently, a collaborative team led by Prof. Huilin Pan (Zhejiang University), Prof. Yang Sun (Sun Yat-sen University), and Dr. Kang Xu (SES AI) published a breakthrough unifying framework in the top-tier journal JACS: Sequenced Interfacial Chemistry. By introducing a binary oxide (MgO), they successfully enforced "traffic rules" upon the interfacial micro-environment.

Spatial & Energetic Regulation: Building an Ordered Micro-environment
Because of the random thermal motion of solvated ions and solvent molecules in liquid electrolytes, local ionic ordering is easily disrupted. The research team innovatively addressed this by coating a polyolefin separator with a simple binary metal oxide (e.g., Magnesium Oxide, MgO@PP).
Precise Interactions: The oxygen (O²⁻) and magnesium (Mg²⁺) sites on the MgO surface exhibit nearly equal-strength interactions with Li⁺ and TFSI⁻ anions in the electrolyte.
Assembling Contact Ion Pairs (CIPs): This balanced interaction acts like an "invisible hand," forcibly capturing and aligning previously free-floating ions into well-defined Contact Ion Pairs at the interface. The initially chaotic interface is structurally and chemically restructured into a highly ordered micro-environment in both spatial and energetic dimensions.

Temporal Decoupling: Making Reactions "Wait in Line"
If space is ordered, the sequence of reactions over time must also be methodical. The researchers used Distribution of Relaxation Times (DRT) to deconvolute the fundamental steps of the interfacial processes.
They found that in unmodified systems, the time scales of various reaction steps overlap severely (indicating high disorder, or "D"). However, in the MgO@PP system, the tangled multi-step reactions (such as desolvation and charge transfer) are clearly separated into discrete, sequential steps. This temporal decoupling dramatically lowers the reaction barrier and interfacial impedance, ensuring that the chemical reactions remain coherent and dynamically stable over continuous cycling.


Lattice Templating & "Catalyzed" SEI: Perfect Deposition
The benefits of MgO extend well beyond regulating the liquid phase; it plays a core role in lithium crystallization and SEI formation:
Isomorphic Lattice "Template": Both MgO and Li⁰ share a cubic crystal structure, and their (200) facets are highly matched. Li⁺ preferentially adsorbs onto the O²⁻ sites on the MgO surface and nucleates along this template. This essentially lays down "train tracks" for the lithium ions, achieving rapid, uniform, and dendrite-free deposition.

Altering the Anion Decomposition Pathway: Traditionally, TFSI⁻ decomposition requires breaking the high-energy N–S bond, resulting in incomplete reactions. Ab initio molecular dynamics (AIMD) simulations reveal that the oxygen sites on the MgO surface inject electron density into the adsorbed TFSI⁻, specifically weakening the S–C bond instead. This critical "assist" redirects the decomposition pathway, allowing the anion to completely and rapidly (within 1258 fs) convert into a robust, inorganic-rich SEI layer (LiF, Li₂O, Li₂S, etc.).
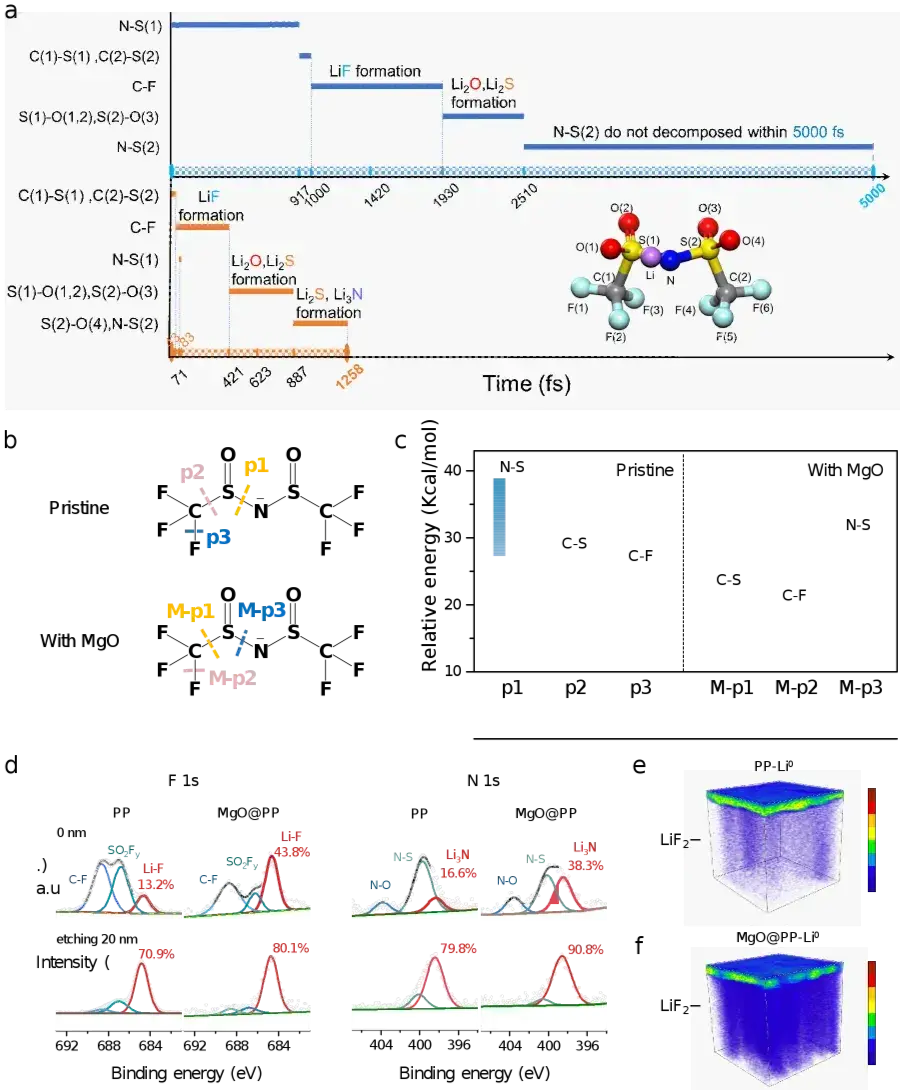
Summary & Outlook
This research establishes a highly universal and elegant paradigm: "Interfacial Ordering." By utilizing a simple binary oxide (MgO), the team successfully reorganized chaotic, random multi-step reactions into a coherent, tightly regulated reaction sequence across time, space, and energy. This provides a unified physicochemical theoretical framework for stabilizing highly reactive metal anodes in next-generation batteries.






Comments